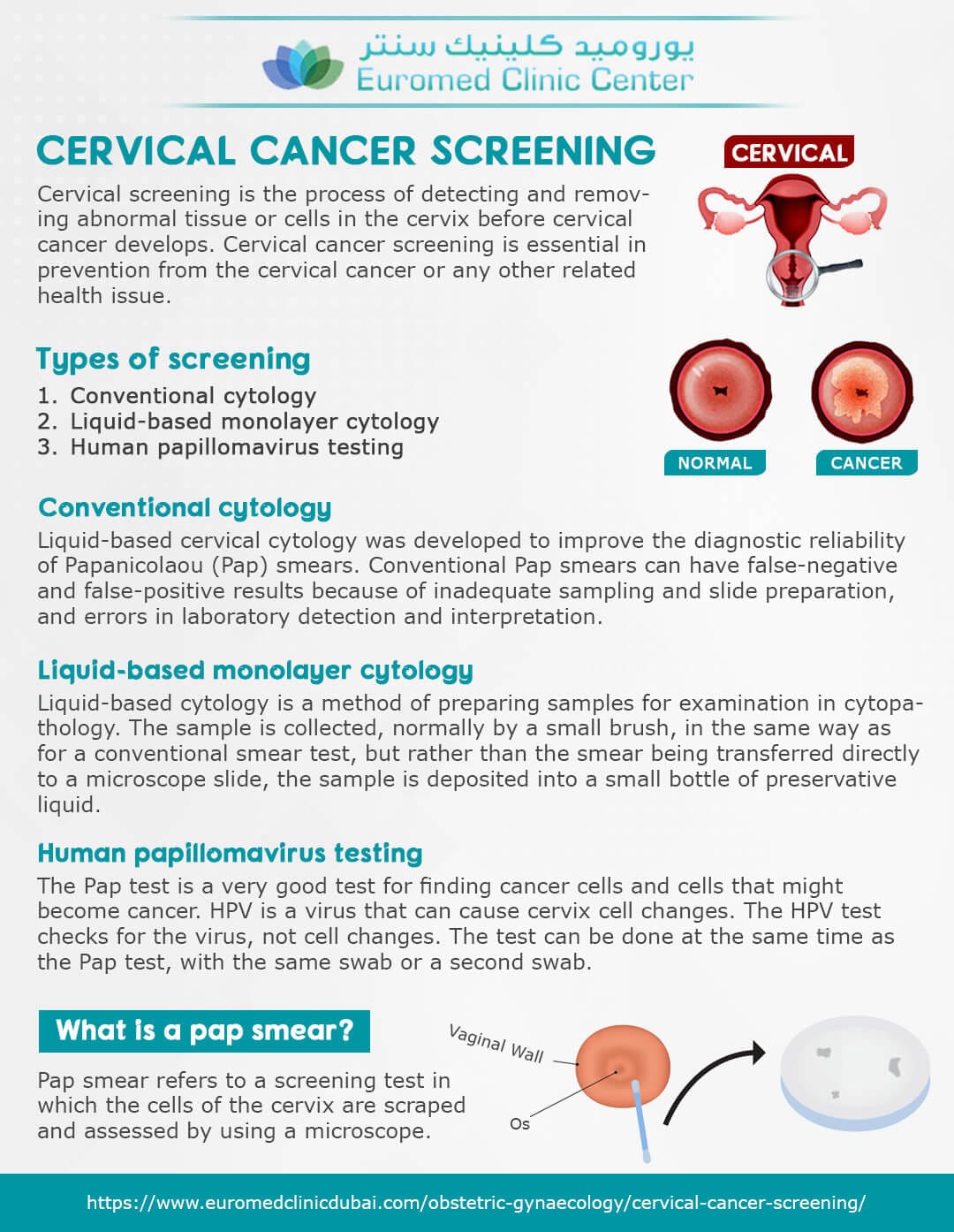
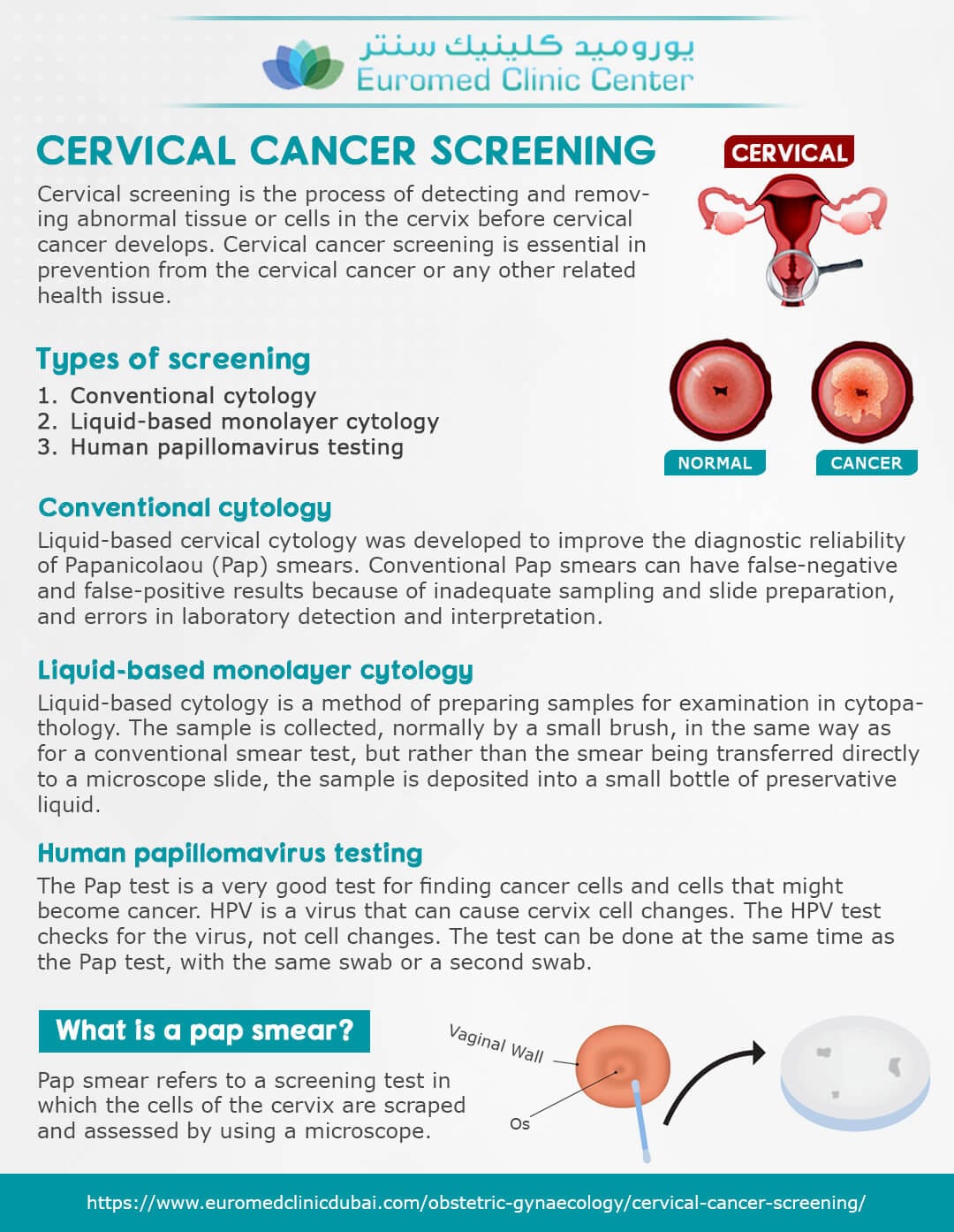
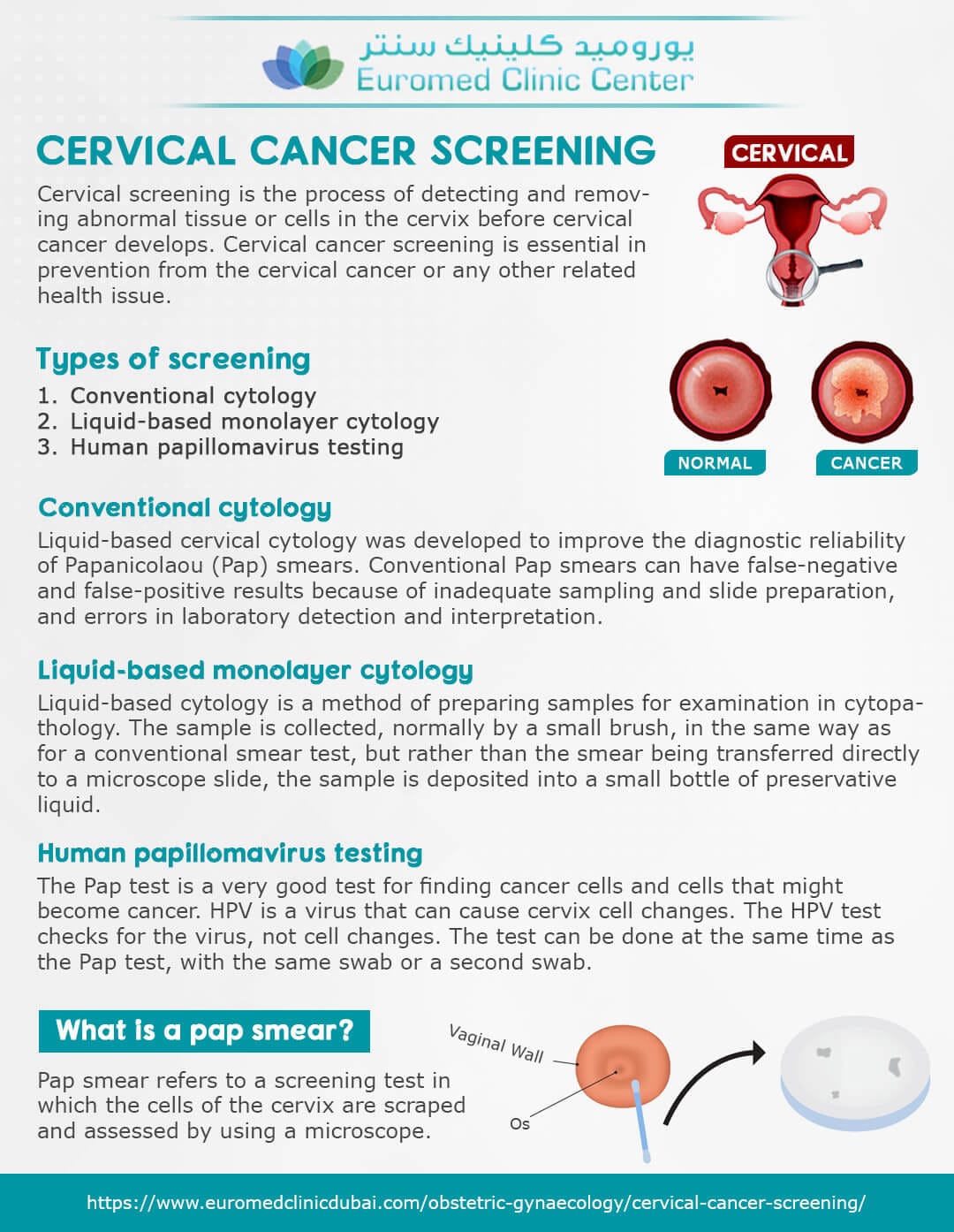
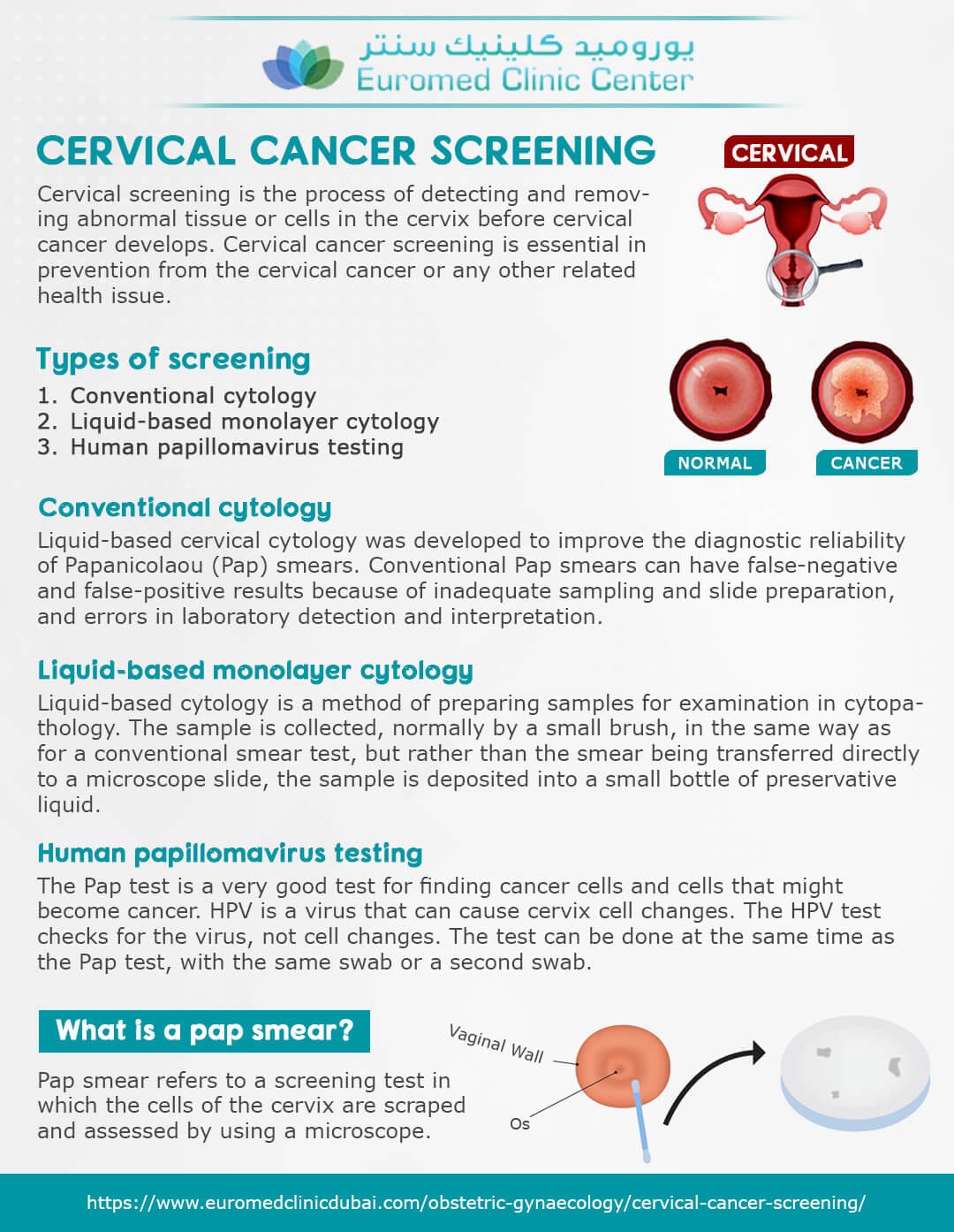
The FDA has officially approved the first cervical cancer screening device designed for home use, offering a new layer of accessibility and autonomy for women’s health. This groundbreaking development is poised to transform how cervical cancer screening is approached, making it more convenient while potentially increasing screening rates.
Tag: cervical cancer
First-of-Its-Kind At-Home Cervical Cancer Screening Device Secures FDA Approval
The FDA has granted approval for a groundbreaking at-home cervical cancer screening device, promising enhanced accessibility for women. With the aim of increasing early detection, this innovative device marks a significant advancement in women’s health technology.
Cervical Cancer Screening Guidelines Shift: HPV Testing Favored for Those 30 and Older, Task Force Suggests
A draft recommendation from a leading health task force proposes a change in cervical cancer screening protocols, suggesting primary HPV testing over traditional Pap smears for individuals aged 30 and older. This shift is based on evidence indicating that HPV testing is more effective at identifying women at risk for cervical cancer. The proposed change aims to improve early detection rates and reduce the incidence of the disease, though Pap testing may remain relevant for certain individuals and circumstances. The task force is currently seeking public comment on the draft.
Cervical Cancer Screening Guidelines Shift Towards HPV Testing
The US Preventive Services Task Force (USPSTF) has drafted a new recommendation that favors HPV testing over Pap smears for cervical cancer screening in women aged 30 and above. This change aims to improve early detection and prevention of cervical cancer.
Draft Recommendation Suggests HPV Testing Preferred Over Pap Smears for Cervical Cancer Screening at 30 and Above
A recent draft recommendation by a task force suggests that women ages 30 and above should prefer HPV (human papillomavirus) testing over traditional Pap smears for cervical cancer screening. This change in recommendation could potentially impact screening guidelines for millions of women across the US and steer them towards more effective and specific tests for early detection of cervical cancer.
Updated guidelines recommend HPV testing over Pap smears for cervical cancer screening beginning at age 30
New guidelines from leading task forces suggest switches from traditional Pap smears to more effective HPV tests for cervical cancer screening, starting at age 30.