In a significant advancement for women’s health, the U.S. Food and Drug Administration (FDA) has approved the first cervical cancer screening device that can be used in the comfort of one’s home. This groundbreaking device, developed by a leading health technology company, offers a novel approach to cervical cancer screening, aiming to enhance accessibility and empower women by providing them with more control over their health.
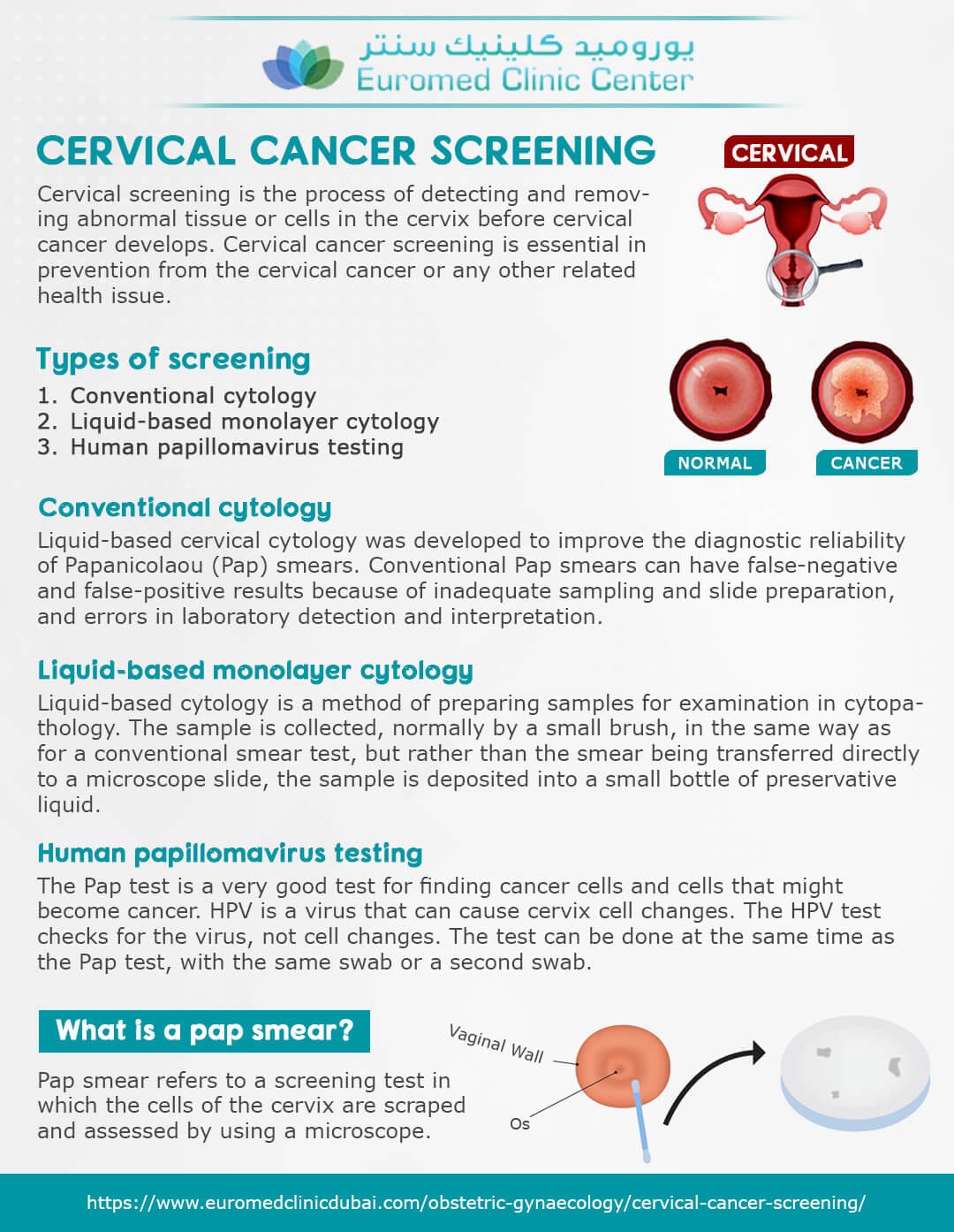
Cervical cancer is a preventable disease, yet it continues to affect thousands of women each year. The primary method of detection is the Pap test, typically performed in a clinical setting. However, traditional screening often presents barriers such as accessibility to health services, time constraints, and privacy concerns. Recognizing these challenges, the FDA’s recent approval represents a pivotal shift towards making cervical cancer screening more user-friendly and widely available.
The at-home cervical cancer screening device operates on a simple yet sophisticated principle. It allows women to collect their own samples with minimal instruction, making the screening process private and less intimidating. After the sample collection, the device can be sent to a lab for testing, where results are processed and communicated back to the user. Such an approach not only increases convenience but can also lead to timely detection of potential issues, ultimately improving health outcomes.
The approval follows extensive clinical trials that demonstrated the device’s efficacy and reliability. Results indicated that the device successfully identified abnormal cervical cells similarly to the conventional Pap test. This is particularly significant in a healthcare landscape where technological advances are increasingly bridging the gap between patients and essential medical services.
Healthcare advocates have long emphasized the need for innovations like this one to promote higher screening rates. According to the Centers for Disease Control and Prevention (CDC), many women skip routine Pap tests due to various reasons, including lack of insurance, long wait times for appointments, and discomfort associated with in-office examinations. The introduction of a home-use device could help reduce these barriers and encourage more women to participate in regular screenings.
Furthermore, the implications of this approval extend beyond just patient autonomy. By enabling women to take charge of their health, there could be a substantial impact on public health and the healthcare system overall. Improved screening rates are likely to lead to earlier detection of cervical cancer, which is critical for effective treatment and successful outcomes.
In addition to enhancing accessibility, the at-home screening device contributes to a larger conversation about technology’s role in healthcare. Digital health tools such as telemedicine have already changed how patients interact with healthcare providers. The introduction of home diagnostic devices represents another step forward, indicating a trend towards more decentralized and personalized healthcare solutions.
However, while the device heralds many positive aspects, it is crucial to approach its use with appropriate education and guidance. Ensuring that women understand how to use the device correctly is essential for accurate results. Therefore, the manufacturer has committed to providing comprehensive instructional materials and support.
Healthcare professionals underscore the importance of following up on any abnormal results. Women using the device are encouraged to consult their healthcare providers for further testing or evaluations if needed. The at-home screening does not replace regular check-ups or healthcare appointments, but rather complements them by providing an additional layer of testing and convenience.
As the approval continues to garner attention, the hope is that it will pave the way for further innovations in women’s health technology. There are already discussions about developing similar home-use devices for other types of cancer screenings, indicating a promising future for broader applications of such technologies.
In conclusion, the FDA’s approval of the first at-home cervical cancer screening device marks a transformative moment in women’s healthcare. By improving accessibility and encouraging proactive health management, this innovative approach could lead to a significant increase in screening rates and, ultimately, healthier outcomes for women. As more women become empowered to monitor their cervical health, the potential to alter the landscape of cervical cancer detection grows exponentially. The healthcare community will continue to watch for developments surrounding this device, as it may very well serve as a model for future innovations in preventive health.
With this approval, the health technology company responsible for the device has taken a monumental step not just for their organization, but for women’s health initiatives everywhere.